
The last steps of this process occur in the mitochondria. The complete breakdown of glucose in the presence of oxygen is called cellular respiration. In either case the PMF needs to be about 50 kJ/mol for the ATP synthase to be able to make ATP. In mitochondria the PMF is almost entirely made up of the electrical component but in chloroplasts the PMF is made up mostly of the pH gradient. Together the electrochemical gradient of protons is both a concentration and charge difference and is often called the proton motive force (PMF). The pH gradient is a result of the H+ ion concentration difference. This gradient is composed of both the pH gradient and the electrical gradient. This charge difference results in an electrochemical gradient. Moving the protons to the outer parts of the mitochondrion creates a higher concentration of positively charged particles, resulting in a slightly positive, and slightly negative side (then electrical potential gradient is about -200 mV (inside negative). In mitochondria free energy released from the electron transport chain is used to move protons from the mitochondrial matrix to the intermembrane space of the mitochondrion. In most cases the proton motive force is generated by an electron transport chain which acts as both an electron and proton pump, pumping electrons in opposite directions, creating a separation of charge. The chemical potential energy refers to the difference in concentration of the protons and the electrical potential energy as a consequence of the charge separation (when the protons move without a counter-ion). This can be described as the storing of energy as a combination of a proton and voltage gradient across a membrane. In all cells, chemiosmosis involves the proton-motive force (PMF) in some step. Ĭhemiosmotic coupling is important for ATP production in chloroplasts Īnd many bacteria. Eventually the weight of evidence began to favor the chemiosmotic hypothesis, and in 1978, Peter Mitchell was awarded the Nobel Prize in Chemistry.  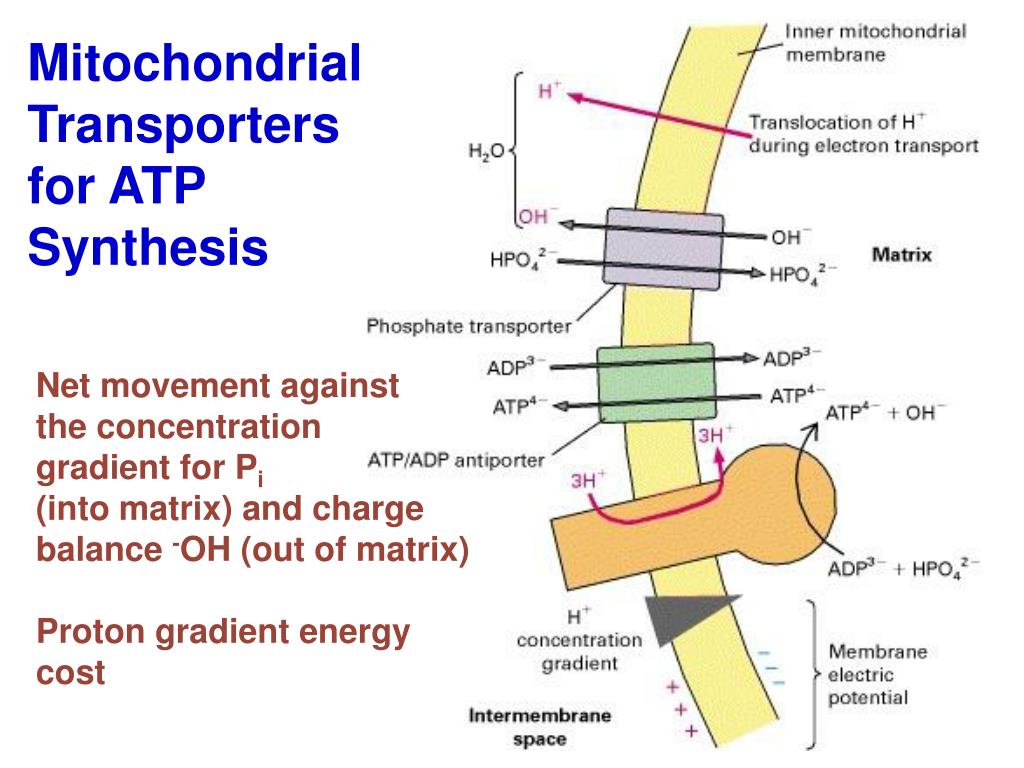 
The problem with the older paradigm is that no high energy intermediate was ever found, and the evidence for proton pumping by the complexes of the electron transfer chain grew too great to be ignored. The prevailing view was that the energy of electron transfer was stored as a stable high potential intermediate, a chemically more conservative concept. This was a radical proposal at the time, and was not well accepted. The electrons and protons at the last pump in the ETC are taken up by oxygen to form water. The flow of protons back into the matrix of the mitochondrion via ATP synthase provides enough energy for ADP to combine with inorganic phosphate to form ATP. The protons move back across the inner membrane through the enzyme ATP synthase. The energy available in the electrons is used to pump protons from the matrix across the inner mitochondrial membrane, storing energy in the form of a transmembrane electrochemical gradient. The carriers pass electrons to the electron transport chain (ETC) in the inner mitochondrial membrane, which in turn pass them to other proteins in the ETC. The oxidation of acetyl CoA in the mitochondrial matrix is coupled to the reduction of a carrier molecule such as NAD and FAD. Molecules such as glucose are metabolized to produce acetyl CoA as an energy-rich intermediate. The theory suggests essentially that most ATP synthesis in respiring cells comes from the electrochemical gradient across the inner membranes of mitochondria by using the energy of NADH and FADH 2 formed from the breaking down of energy rich molecules such as glucose. Mitchell proposed the chemiosmotic hypothesis in 1961.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
